Recently, results of the Asian subgroup in ASTRUM-004, Henlius’ pivotal phase 3 clinical study of anti-PD-1 mAb HANSIZHUANG (serplulimab) plus chemotherapy as first-line treatment for previously untreated locally advanced or metastatic squamous non-small-cell lung cancer (sqNSCLC), were released at European Society for Medical Oncology Asia (ESMO Asia) Congress 2023 in poster presentations. ASTRUM-004 was led by Professor Caicun Zhou from Shanghai Pulmonary Hospital and its results were initially presented at the IASLC 2023 World Conference on Lung Cancer (WCLC 2023) in the form of oral presentation, showing that serplulimab significantly improved survival with a manageable safety profile in previously untreated locally advanced or metastatic sqNSCLC patients. Previously in 2022, the new drug application (NDA) of serplulimab for the treatment of sqNSCLC has been approved by the National Medical Products Administration (NMPA) based on the results of ASTRUM-004.
HANSIZHUANG is Henlius’ first innovative product, as well as the first and only anti-PD-1 therapy approved for the treatment of small cell lung cancer (SCLC). It has been approved by the NMPA for the treatment of MSI-H solid tumours, squamous non-small cell lung cancer (sqNSCLC), extensive-stage small cell lung cancer (ES-SCLC), and esophageal squamous cell carcinoma (ESCC). ASTRUM-004 is a randomized, double-blind, international multicenter phase 3 study to compare the clinical efficacy and safety of serplulimab plus chemotherapy versus chemotherapy in patients with locally advanced or metastatic sqNSCLC who have not previously received systemic treatment. The study was conducted in various countries, including China, Poland, and Turkey, and enrolled 537 patients.
Data from its Asian subgroup released at 2023 ESMO Asia are as follows:
Title
Phase 3 study of serplulimab plus chemotherapy as first-line therapy for advanced squamous non-small-cell lung cancer: ASTRUM-004 Asian subgroup
Study design
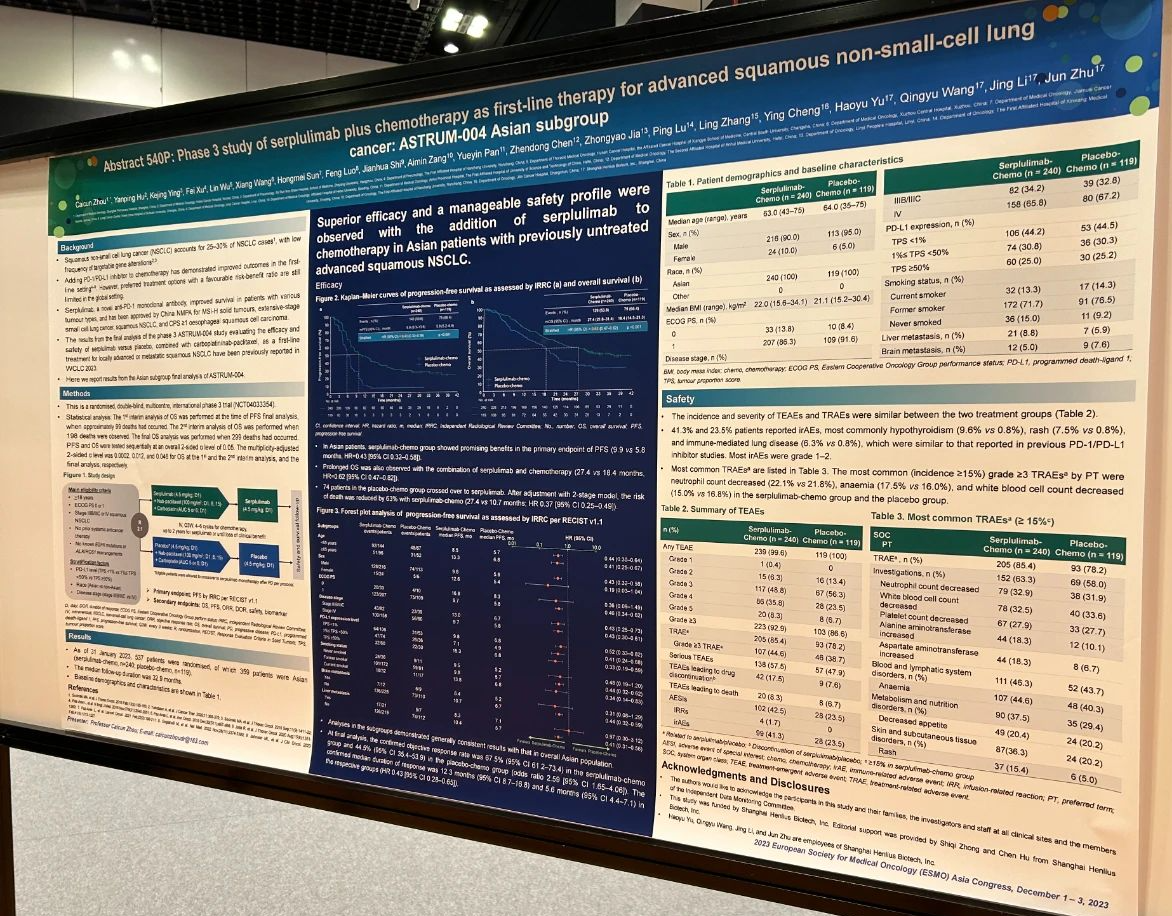
Patients with stage IIIB/IIIC or IV squamous non-small-cell lung cancer (sNSCLC) and no prior systemic therapy were randomised 2:1 to receive serplulimab 4.5 mg/kg or placebo (up to 35 cycles) plus chemo (carboplatin/nab-paclitaxel, 4–6 cycles) in 3-week cycles. Randomization was stratified by PD-L1 expression level (TPS ≥50% vs. 1%≤ TPS <50% vs. TPS<1%), race (Asian vs. non-Asian), and disease stage (stage IIIB/IIIC vs. IV). Primary endpoint was IRRC-assessed PFS per RECIST 1.1.
Results
Primary endpoint was met in the overall population. As of 31 January 2023, 359 Asian patients were randomised (serplulimab-chemo, n=240; placebo-chemo, n=119). With a median follow-up of 32.9 months, IRRC-assessed median PFS was longer in serplulimab-chemo group than in placebo-chemo group (9.9 vs. 5.8 months; HR 0.43, 95% CI 0.32–0.58). The HR for PFS consistently favoured serplulimab-chemo group regardless of PD-L1 expression level or disease stage. OS was prolonged with the addition of serplulimab (median, 27.4 vs. 18.4 months; HR 0.62, 95% CI 0.47–0.82). 41.3% and 23.5% patients reported immune-related adverse events (irAEs), most commonly hypothyroidism (9.6% vs. 0.8%), rash (7.5% vs. 0.8%), and immune-mediated lung disease (6.3% vs. 0.8%). Most irAEs were grade 1–2.
Conclusion
Superior efficacy and a manageable safety profile were observed when serplulimab was added to chemo in Asian patients with untreated advanced sNSCLC. Serplulimab plus chemo has been approved in China and maybe recommended for this patient population.
About HANSIZHUANG
HANSIZHUANG (recombinant humanized anti-PD-1 monoclonal antibody injection, generic name: serplulimab injection) is the first anti-PD-1 mAb for the first-line treatment of SCLC. Up to date, 4 indications are approved for marketing in China, 1 marketing application is under review in the EU, and more than 10 clinical trials are ongoing across the world.
HANSIZHUANG was launched in March 2022 and has been approved by the NMPA for the treatment of MSI-H solid tumours, squamous non-small cell lung cancer (sqNSCLC), extensive-stage small cell lung cancer (ES-SCLC), and esophageal squamous cell carcinoma (ESCC). Its marketing applications of the first-line treatment for ES-SCLC are under review by the EMA. Focusing on lung and gastrointestinal cancer, the synergy of HANSIZHUANG with in-house products of the company and innovative therapies are being actively promoted. It has successively obtained clinical trial approvals in China, the U.S., the EU and other countries and regions to initiate more than 10 clinical trials on immuno-oncology combination therapies in a wide variety of indications. As of now, the company has enrolled more than 3,600 subjects in China, the U.S., Turkey, Poland, Georgia and other countries and regions, and the proportion of White is over 30% in two MRCTs, making HANSIZHUANG an anti-PD-1 mAb with one of the largest global clinical data pools. The results of 3 pivotal trials of HANSIZHUANG were published in the Journal of the American Medical Association (JAMA), Nature Medicine and the British Journal of Cancer, respectively. Furthermore, HASIZHUANG was recommended by the CSCO Guidelines for Small Cell Lung Cancer, the CSCO Guidelines for Non-Small Cell Lung Cancer, the CSCO Guidelines for Esophageal Cancer, the CSCO Guidelines for Colorectal Cancer, the CSCO Clinical Practice Guidelines on Immune Checkpoint Inhibitor, the China Guidelines for Radiotherapy of Esophageal Cancer, and other definitive guides, providing valuable references for clinical diagnosis and treatment of tumours. On the other hand, serplulimab was granted orphan drug designations by the U.S. FDA and the EC for the treatment of SCLC, and its bridging head-to-head trial in the United States to compare HANSIZHUANG to standard of care atezolizumab (anti-PD-L1 mAb) for the first-line treatment of ES-SCLC is well under way.
