Shanghai, China, Febuary 1st, 2024-Shanghai Henlius Biotech, Inc. (2696.HK) announced that a phase 1 clinical trial of the company’s denosumab biosimilar HLX14, a recombinant anti-RANKL human monoclonal antibody injection, met its primary endpoint. The results of this study suggest that HLX14 had highly similar PK and PD, as well as comparable safety, tolerability, and immunogenicity to the reference drugs from different sources. This study met all the pre-specified endpoints. The company is also actively conducting an international multicentre phase 3 comparative clinical study (NCT05352516) of HLX14 in postmenopausal women with osteoporosis at high risk of fracture.

Henlius independently developed denosumab biosimilar HLX14 in accordance with the NMPA, EMA, FDA, and other international biosimilar guidelines. This is a two-part phase 1 clinical study in Chinese healthy adult male subjects. Part 1 is an open-label, randomised, parallel-controlled, single-dose, two-arm pilot study with the primary objective to compare the PK parameters of HLX14 and EU-sourced denosumab to provide further basis for the study design of part 2. The secondary objective of part 1 is to compare the PD, safety, tolerability, and immunogenicity of HLX14 and EU-sourced denosumab. Part 2 is a double-blind, randomised, parallel-controlled, single-dose, four-arm study with the primary objective to compare the PK similarity of HLX14 with US-, EU-, and CN-sourced denosumab ("reference drug"). The secondary objective of Part 2 is to compare PD, safety, tolerability, and immunogenicity between HLX14 and the reference drugs. The results of this study suggest that HLX14 had highly similar PK and PD to the reference drugs from different sources, with geometric mean ratios (GMRs) of primary PK endpoints near 1 and their 90% confidence intervals (CIs) falling entirely in pre-specified equivalence margins of 0.8 to 1.25. Safety, tolerability, and immunogenicity were also comparable. This study met all of the pre-specified endpoints.
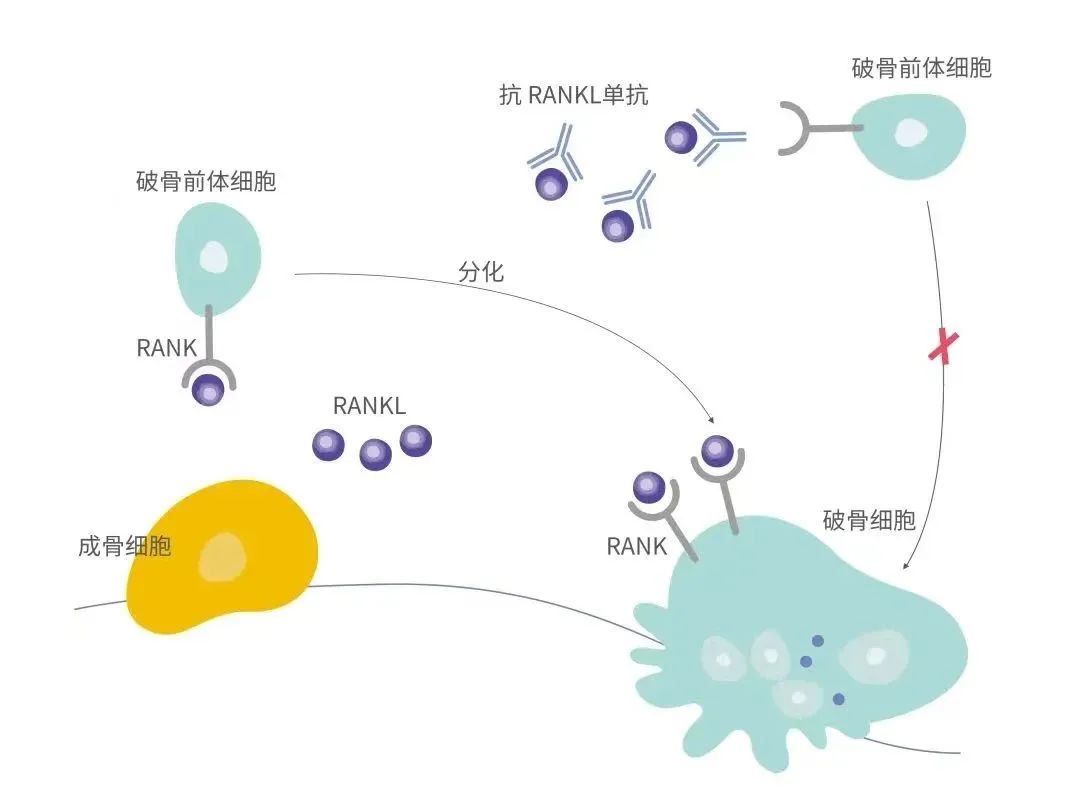
HLX14 can specifically bind to RANKL (receptor activator of nuclear factor kappa B ligand) and block the interaction between RANKL and RANK, which is expressed on the surface of osteoclasts, thus inhibiting RANKL/RANK-mediated differentiation, maturation and activation of osteoclasts, thereby reducing bone resorption and the incidence of skeletal-related events [1]. Denosumab has been approved in various countries and regions for a range of indications such as for the treatment of osteoporosis in postmenopausal women at high risk for fracture, osteoporosis in men at high risk for fracture, bone loss in specific populations at high risk for fracture, prevention of skeletal-related events in patients with bone metastases from solid tumours and in patients with multiple myeloma, and the treatment of giant cell tumour of bone and hypercalcemia of malignancy, etc.
In 2022, Henlius entered into a license and supply agreement with Organon LLC for the exclusive commercialization of two biosimilar candidates, including HLX14, in ex-China countries, covering mature markets such as the United States, the European Union and Japan, as well as a number of emerging markets. Henlius will always place a high value on patients’ needs and clinical data and will continue to diversify innovation by strengthening internal innovation capabilities and external collaboration, extending new drug forms, and processing more clinical trials, to benefit patients around the world.
【参考文献】
[1]. Romas E. Clinical applications of RANK-ligand inhibition. Intern Med J 2009;39:110–6.
